European Badger Breeding

Mating may occur during any month, suggesting that badgers may be induced ovulators - i.e. it is the act of mating that triggers ovulation, rather than seasonal factors such as day length or temperature. Nonetheless, both field observations and histological data suggest the majority of mating happens in two peaks: the first, and largest, is between January and May, with the bulk of that in early Spring (i.e. February and March); the second during July to September. Histological studies on the uteri of dead badgers suggests that females can ovulate in any month except December, with yearlings about two months later in their peaks than older animals. Conventional wisdom suggests boars are fertile throughout the year in good habitats, those in harsh or poor quality habitat being typically infertile during winter, although preliminary data from Oxford University's WildCRU research centre suggests males may also experience peaks in fertility, with some stopping sperm production after the spring mating peak and others remaining fertile into autumn. Oestrous (the female's receptive period) is between four and six days, which means boars have a relatively narrow window of opportunity. The vast majority of courtship behaviour appears to happen at the sett, but much may also take place underground. Consequently, we know relatively less about this behaviour than we do for other mammals.
A common features of courtship include a "shuffling" or stiff-legged approach by the boar, soft vocalisation and, frequently, the male chasing the sow. In their 1996 edition of Badgers, Ernest Neal and Chris Cheeseman describe some mating events:
"The first sign of interest shown by the boar is often the raising of the tail into a vertical position and the emission of a loud and often continuous deep whinnying purr. He may then approach with a shuffling motion taking short steps with the legs kept rather rigid."
Other descriptions relate the presence of mutual grooming and "ground pawing" by the boar immediately prior to mating and/or a curious behaviour during which the sow runs in circles, first clockwise, then anti-clockwise, prior to mating, although it is unknown how common this latter activity is. The male may also scent mark the ground, scraping the earth to the sett entrance, and, in his 2010 book Badgers, Tim Roper described the emission of anal gland scent associated with copulation being sufficiently potent that "it is still evident to the human nose several hours later". Many field reports describe vocalisation, particularly deep rumbling "churring" by the boar to entice the sow from the sett and "purring" during approach and copulation, during copulation. Interestingly, the sow may also initiate copulation by raising her tail and backing onto the boar's flanks, which prompts him to raise his tail and mount her.
Copulation may happen as soon as the sow leaves the sett or after a protracted period of courtship/foreplay with serious attempts lasting anywhere from ten minutes to an hour and a half. Mating bouts of less than five minutes are often taken to indicate the sow isn't fully receptive, although copulation length varies with season, the longest bouts during February and March. The boar mounts the female from behind, grabbing her waist with his front paws and biting her nape. Copulation may be terminated by either party, the male dismounting after ejaculation or the sow twisting to get free or, in one description, walking under a fence to push the boar off.
All for one?
Badgers are promiscuous and both sexes will mate with individuals from other clans. In a fascinating short paper to the Journal of Zoology during 1995, University of Sussex zoologist Sean Christian described a boar mating with a sow from a neighbouring sett while the resident male was away and also mate-guarding, where the resident male fought with and subsequently chased intruding males from the territory while the resident sow was in oestrous. While it may be risky for males to intrude looking for sows, females appear able to move into neighbouring territories where they're free to mix with, and frequently mate with, other badgers.
In a 2007 paper to the journal Molecular Ecology, a team of biologists led by Hannah Dugdale at the University of Oxford reported on the genetic makeup of the badgers in Woodchester Park, Gloucestershire. They found that males from outside of the sow's social group, typically boars from neighbouring social groups, sired about half of the cubs to which they could confidently assign paternity. Moreover, there were very few (~22%) successful matings between members of the same clan. These cases of observed and inferred promiscuity undoubtedly serve to reduce the threat of inbreeding. Indeed, a paper published in the Quarterly Review of Biology suggests that the delayed implantation coupled with superfetation may combine to help increase the reproductive fitness of female badgers.

Superfetation is a condition whereby two foetuses, fertilised at different times, are present in the uterus at the same time. Despite claims that many animals exhibit superfetation, verifiable cases are rare and the condition has only been well documented in the group of freshwater fish called mollies (Poeciliidae). The authors of this study argue that this combination of superfetation and embryonic diapause may make it difficult for dominant boars to identify cubs that are sired by other males, promoting what the biologists refer to as "cryptic polyandry". Not only does this cryptic polyandry reduce the likelihood of inbreeding, but it may also reduce the probability of infanticide -- dominant badgers are known to kill the offspring of subordinates -- and increase the breeding sow's window of reproductive opportunity by increasing her scope for conception.
Delayed implantation
In a study of 548 adult female badgers from Offaly in Ireland between 1989 and 1990, Thomas Hayden and Robert Whelan found that between 80% and 90% of females mated, with 65-70% achieving successful implantation and, of these, only 35-40% lactated. These figures are similar to results from studies on badger populations in southern England, where about one-third of females breed annually. The Irish paper goes on to report that reproductive performance of yearlings was inferior to older badgers and observed some evidence for density-dependant reproduction -- i.e. reproductive success was correlated with the number of badgers in the population. This concept is also something Neal and Cheeseman consider during their summary of implantation rates across Europe in Badgers, noting "It would be interesting to know what happens in regions where badgers are rare and social groups restricted, perhaps to a single pair. An even higher success rate would appear to be probable." In Britain, it seems most badgers will implant during mid-December in southern England, late December in northern England and early January in Scotland, with those in good body condition implanting earlier than those with lower fat reserves.

After fertilization, sows undergo a phenomenon known as embryonic diapause or delayed implantation. Diapause is a dramatic reduction in, or a cessation of, mitosis in the zygote at the blastocyst stage. In other words, the cells of the fertilized egg multiply to form a hollow ball (called a blastula) and then stop, remaining suspended in the uterus for between three and 15 months. Currently, reproductive biologists divide those species displaying embryonic diapause into those which are facultative (i.e. delayed implantation is induced by environmental conditions, such as in the rodents and marsupials) and those that are obligate (i.e. that present at every gestation regardless of ambient conditions). Badgers undergo obligate diapause; sows delay implantation until late December or early January, regardless when mating occurs.
In 1880, S. Fries proposed that delayed implantation may allow badger cubs to be born earlier in the growing season, thus providing them with more time to develop before they must face their first winter. Some 130 years on, Fries' hypothesis is still considered the most plausible explanation for this phenomenon. Indeed, in their paper to the journal Evolution, Michael Thom, Dominic Johnson and David Macdonald of Oxford University argued that delayed implantation is a plesiomorphic (i.e. primitive) characteristic in mustelids, which evolved several times, probably because there are costs associated with it. The observation that delayed implantation increases in frequency as you move further from the equator, argue the zoologists, strongly supports the idea that it provides the beneficial trait of being able to uncouple mating and parturition. In other words, diapause permits sows the luxury of being able to select mates at premium times (e.g. when males have survived a particularly harsh season, suggesting high quality) while allowing births to coincide with the most favourable seasons. Having three cubs born at the same time, despite having been conceived at different times and potentially by different fathers, also increases the potential genetic diversity of the litter, making it more likely at least one cub will survive to adulthood.
The biochemical cause of this diapause phenomenon has yet to be established. It has been suggested that hormones in the badger's blood (e.g. progesterone and oestrogen) and possibly some proteinacious development factors are responsible for triggering resumption of blastocyst development. Indeed, studies in rodents have suggested that the female sex hormone oestradiol and Epidermal Growth Factor Receptor (Egfr) may play a role in triggering blastocyst implantation; certainly, a single injection of the female sex hormone oestrogen has been shown to terminate diapause in some species. Similarly, work by Rene Canivenc at the Universite de Bordeaux II in France has demonstrated a pronounced increase in luteal vascularization and progesterone production prior to implantation in badgers.

In April 1979, a paper published in Nature reported on the experiments of Rene Canivenc and colleague M. Bonnin on captive sows. The biologists exposed six sows to a 10:14 photoperiod (i.e. 10 hours of light, followed by 14 hours of darkness) at 5oC (41oF). Blood samples were taken from the badgers each week and analysed for progesterone, a steroid hormone secreted by the corpus luteum to prepare and maintain the uterus for pregnancy. The scientists found that when the photoperiod was decreased (i.e. more dark, less light) progesterone levels rose to those seen just prior to implantation during January in wild specimens (about 20ng per mL).
Through manipulation of the photoperiod, Canivec and Bonnin could induce "whelping" (i.e. birth) during September, while in the wild the litter would not have been born until the following spring. Ultimately, Canivenc and Bonnin succeeded in demonstrating that delayed implantation in the Eurasian badger appears to be under environmental, rather than genetic, control. Subsequent experiments by the same authors, during which the temperature was controlled, suggested it was light, rather than temperature, that triggered implantation.

These experiments give us a good idea of some of the factors at play here, but they don't appear to tell the whole story. As Neal and Cheeseman point out, there are several questions that have yet to be answered. Among the problems is the fact that from about October-time onwards, badger emergence is entirely nocturnal and not obviously correlated with sunlight. In addition, we'd expect implantation to be earlier at northern latitudes, but we actually see the opposite. Finally, there are (admittedly rather rare) records of two sows living in the same sett giving birth several weeks apart.
Data from Stephen Ferguson and colleagues at the University of Saskatchewan in Canada suggest that reduced nutrition in the sow can lead to a lengthening of the diapause period. More recent work seems to support this idea and an interesting experiment by a team at the University of Oxford found that when females had a relatively high body condition index (i.e. were healthy and well fed), they implanted early and the cub sex ratio was male biased (i.e. more males were born than females). The important point here is that their finding goes against the reasonably well-established Local Resource Competition Hypothesis (LRCH). The LRCH states that in years when female body condition is poor animals should lower competition for local resources by producing males, which are more likely than females to disperse.
The connection between fat and hormones does seem to be an important one and Ernest Neal speculated that, given the lipophilic tendencies of steroids (i.e. they are fat soluble), hormones may be incorporated into the fat reserves built up during the summer and autumn. When the sow enters winter torpor, she begins to live off her fat reserves and as the fat is metabolised, the hormones are gradually released into the bloodstream; it may be this process that triggers implantation. There are various lines of indirect evidence that seem to suggest this is a plausible theory and, if correct, would make the rather inactive month of December a crucial time for the triggering of blastocyst implantation. Indeed, field observations seem to indicate that disturbed setts produce fewer litters than undisturbed ones.
When we consider the foregoing, in combination with reports from the field suggesting litters are closely tied with food availability (i.e. in years of poor food supply cubs are often later than usual), it becomes clear that the trigger for implantation still begs research. Ultimately, it is probably a combination of light, temperature, current body condition and food supply that interact to trigger implantation.
Gestation, birth & development
Once implantation of the blastocyst has occurred, gestation is usually between six and eight weeks, with most cubs born from January to March - based on data from south west England, the bulk of cubs are born during the first half of February, although this peak will likely shift to later in the year as you move north. More generally, birthing dates vary with both latitude and altitude, with more northern badgers and those at higher elevations giving birth later. There are occasional reports of heavily lactating sows in summer and the latest example I have come across in the UK is one killed on a road in Eastwood, Nottinghamshire on 14th August 2020. I have also seen an unverified report of very small cubs found in the Netherlands during August 2019.
Despite most badgers being winter- or spring-born, births have been recorded (or calculated based on estimated cub age at emergence) in most months. In their dataset of 97 litters in southern England, for example, Neal and Cheeseman list three litters born during December, one in early June 1993 and one each during late July and early August 1956 in Devon. In his 1940 opus to Zeitschrift für Jagdkunde on the biology of the badger in Germany, Harry Frank made mention of a tiny cub, with eyes still closed, retrieved from a sett during June. Similarly, a reader contacted me in early July 2020 with a report of a sow visiting her garden who was, based on stomach distention and protruding teats, either heavily pregnant or had very recently given birth. In Ernest Neal's (1986) words, however, births from May to November "can be looked upon as exceptional".
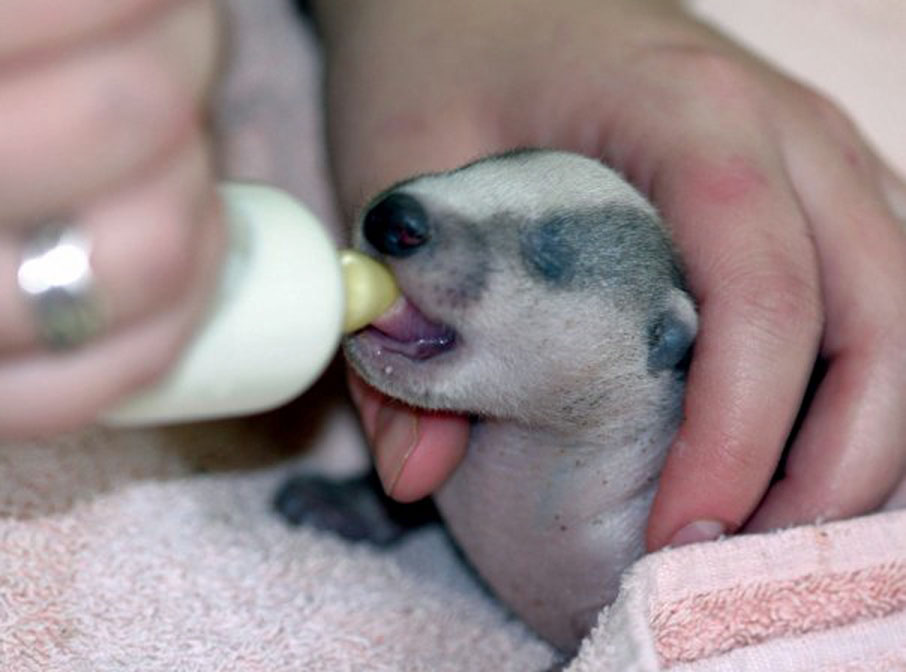
A female may produce as many as six cubs, although two or three appears most common. Studies from Ireland have recorded in utero litter sizes of between one and four young. Typically, any given female will only produce one litter in a single year, although under plentiful conditions more than one female in the clan may reproduce. In common with all mammals, the young are altricial (i.e. born blind and helpless). Neonatal (newborn) badgers are thin with pale grey fur and measure about 12 cm (5.5 in.). The cubs have their first teeth at about four weeks old and their eyes open after about five weeks. The sow will groom the anus of the cubs to stimulate them to urinate and defaecate, as cubs cannot initially evacuate their bowls by themselves. Caroline Gould at Vale Wildlife Hospital has noted that badger cubs seem to take longer to reach this stage than fox cubs, needing help until they're about three weeks old, and sometimes a little longer.
The majority of cubs are born underground in a specially modified chamber close to the sett entrance, with good ventilation and a mass of bedding that is moved in prior to the birthing. Based on captive badger births, it has been suggested that the temperature inside the natal chamber is 18 to 20C (64-68F). In rare instances, a sow may give birth above ground on large mounds of straw/hay/grass/reeds. This appears to be more likely where the expectant mother is a subordinate sow looking for somewhere to give birth away from the attention of the dominant sow. Sows will move their cubs to alternative locations if they feel they are are risk. In the New Forest during 2022, Andrew Walmsley observed a sow moving her cubs twice within about a week. Intriguingly, also in Hampshire, on several occasions during March 2025, Barry Deakin captured an apparently indecisive sow leaving the sett with a cub, before bringing it back anywhere from a few minutes to eight hours later. When she returned, the sow moved towards the sett entrance with the cub in her mouth where she stopped. She turned around and took the cub away again, leaving the frame for a couple of minutes, before reappearing and taking the cub underground. It's unclear what triggered either the initial moving of the cub(s) or the sow's capriciousness.
In common with all mammals, badger cubs are initially dependent on milk produced by the sow, although observations by Gösta Notini in Sweden and Robert Howard and Keith Bradbury in the UK imply adults may also regurgitate food for cubs while underground. Suckling of the cubs almost invariably happens below ground, the female lying on her back or side in a nest chamber, and the sow will return periodically during the night to check on and suckle the cubs. Up until they're about four-weeks-old the cubs lose heat rapidly and the mother doesn't stray far. When the cubs are 6-8 weeks old, the sow will feed away from the sett and return three or four times per night to suckle them, sometimes remaining in the sett for an hour or so before leaving to resume feeding. During periods of prolonged drought, Neal and Cheeseman note that sows may continue to lactate well into the autumn, suggesting badgers can continue suckling even during adverse conditions. I have been unable to find any analysis of the chemical composition of badger milk.
Young badgers emerge from the nursery chamber at about eight weeks old (late April or early May) and begin supplementing their diet with food they find in the vicinity of the sett straight away, thus beginning the weaning process. There are a couple of observations, recounted by Neal and Cheeseman in Badgers, suggesting that the sow may regurgitate food for her cubs during weaning. One particular example, recounted from Chris Ferris' June 1982 diary, described a sow regurgitating "a great heap of worms which the cub proceeded to eat" in response to mouth licking by the excited cub. Unlike foxes, clan members do not bring food back for the cubs, presumably reflecting that the diet of the badger is more omnivorous than the fox and cubs can therefore find more sustenance local to the sett. The permanent dentition is complete by about 16 weeks (ca. 4 months) and the cubs are typically independent by around five months old, at the end of June or beginning of July. Cubs may be seen foraging with the sow by summer and by June the juveniles will be familiar with the clan's territory by this stage.

By the time the cubs are fully weaned the juvenile badger may weigh 6 kg (13 lbs) if conditions are good. At the end of the year it will usually weigh 8-10 kgs (19-22 lbs) and measure 70 to 80cm (2.5 ft.).
Growing up
On average both males and females mature at 12 to 15 months old, although males may mature as late as two years and females may begin ovulating earlier or later than the average. Dispersal is most common in badgers of two years old and seems intrinsically related to clan stability (the more stable the habitat and social dynamics of the clan, the less likely dispersal becomes) and males have a greater tendency to disperse than females.
Studies on the badger population in Oxford's Wytham Woods by Rebecca Fell and colleagues suggest that cubs generally don't have a significant impact on sett society early in their lives, being left either completely alone or accompanied only by their mother. As they grow up, however, they loosen their grip on the "apron strings" and spend increasing amounts of time following and soliciting play from other members of the clan, until they're interacting with most of the group by the age of about four months. As Tim Roper put it in his 2010 book Badgers:
"... cubs undergo a gradual process of social integration in which they themselves, rather than adult members of the group, take the initiative."

While watching a sett in west Yorkshire, Adrian Middleton noted that adults, even the dominant boar, did sometimes play with the cubs - play consisting of chasing, "whickering", wrestling and rolling around. Indeed, former wildlife ranger and rehabilitator Mal Ingham tells me that he frequently sees adults, and particularly boars, playing with cubs at the setts he watches and Dave Sturgess posted some interesting footage to the BBC Springwatch Facebook group in June 2020 showing a boar playing with a cub (grabbing its tail and rolling around) in his garden in Norfolk. Interestingly, Middleton also described play behaviour between adults at the sett.
We know very little about survival of cubs before they emerge from the sett. There are examples of dead cubs, even very young animals, having been found around the sett, although we don't know the story behind their death. Infanticide is certainly a danger for cubs and there are reports in the literature suggesting that dominant boars will kill cubs sired by subordinates or individuals from outside the clan. Additionally, in February 2021, naturalist Kate MacRae captured footage of a large boar who had previously been observed scraping and scent-marking around her artificial sett investigating a nest chamber into which two two-day-old cubs were sleeping. On her blog, Kate described the behaviour of the male:
"Suddenly he saw or heard the cubs in the front, left hand corner. With lightning speed he homed down on them. Grabbing them, he shook them violently and, in just a few seconds, had killed both. He then left the chamber."

The sow returned shortly afterwards and pushed the bodies of the cubs around the chamber briefly before eating one of them. This is the only firsthand account of such infanticide among wild badgers.
Overall, about half the cubs will die within their first year through causes other than infanticide (i.e. the weather, disease, on the roads etc.) and there is an average annual adult mortality of about 30%. One study from Ireland published in 1993 observed that more than 60% of cubs died during their first year, 35% to 40% of which died before they were weaned. Males were also more likely to die early in the year, with females dying later. Presumably this trend reflects the tendency for males to be bolder and thus explore further from the relative safety of the sett than females.
Disease and road accidents can have a significant effect on some populations, but weather is perhaps the most significant mortality factor. In a paper to the Journal of Zoology in 2002, a team from Oxford University's WildCRU lab report on the population dynamics of the badgers living in Wytham Woods between 1987 and 1996. During this time, the population expanded considerably, almost quadrupling from 65 adults to 228, as did badger density (15 adults per sq-km in 1987 to 38 adults per sq-km in 1996).
Despite the substantial population increase, cub survival was inextricably linked to rainfall. In 1990, a summer of unusually low rainfall resulted in 15 (65%) of the year's 23 cubs dying, although rainfall seemingly had little impact on the number of adults surviving. Presumably, during prolonged periods of dry weather, earthworms are scarce and difficult to dig for, leading to a decline in overall body condition and an increased chance of cub mortality. Fortunately for the badgers of Wytham, their population appears to have grown in response to an overall improvement in climatic conditions as well as the social rearrangement of territories observed during the study.