Summary: Rabies is an acutely infectious viral disease of the central nervous system (CNS), which is typically transmitted in the saliva of infected animals, especially dogs; transmission is most commonly via a bite. In foxes, symptoms include seizures, inability to drink (hydrophobia), disorientation, being in a “zombie-like” state and, in some cases, biting at objects and other animals. It should be noted, however, that many of these symptoms are not unique to rabies infection. In humans, early diagnosis with wound cleaning and vaccination following a bite has a good prognosis; once symptoms have manifested, however, rabies is invariably fatal.
Britain has been free of rabies since 1922 and, following a substantial vaccination drive, many western European countries are also now clear of the virus. Rabies is still endemic across much of eastern Europe, however, where the main wildlife carrier is the Red fox. In the USA and Canada, raccoons and Arctic foxes, respectively, are considered the main carriers, although skunks are significant carrier in the US and Red foxes are maintenance hosts in both countries.
Virus variants are known to circulate within species groups (‘fox rabies’ circulates in the fox population, ‘dog rabies’ among dogs, ‘raccoon rabies’ among raccoons, etc.), and the virus tends to not be sustained outside of that group – i.e. a rabid skunk may infect a fox, but the fox is unlikely to infect other foxes. There is, however, sometimes spill-over between closely-related hosts; foxes to other canids, for example. Initially, anti-rabies campaigns involved widespread culling of foxes, but this was costly, labour intensive and largely ineffective – success was achieved in a couple of cases, but typically culling could not suppress fox numbers below the density needed to sustain the virus (one fox per two sq.-kilometres). Culling also maintains fox populations in a state of flux, resulting in more fights as newcomers establish territories, and this can make the situation worse.
The greatest success in eradication has come from a widespread vaccination initiative that began in Switzerland during the late 1970s and stopped the westward progression of the disease; in time, forcing it back eastwards. Baits containing capsules of the vaccine are dropped in infected areas; these are eaten by the resident foxes, which effectively form a ‘living wall’ of immunised animals that prevent the virus getting a foothold. In some countries, rabies is now considered a minor disease and widespread baiting has ceased because the costs now out-weigh the benefits. It remains to be seen whether outbreaks arise if the number of immune individuals drops below the threshold.

Rabies, a Latin word meaning ‘madness’, has the ability to strike fear in the hearts of most people. The origins of this disease are enigmatic, but certainly not recent. Indeed, there is a curious association that some make with a story in Greek mythology; in the fate that befell the hapless hunter Acteon more than a millennium ago. There are many variations on the story, but one -- recounted by David Macdonald in his book Running with the Fox -- tells how Acteon happened upon Diana, Goddess of the Hunt, and her nymphs bathing naked; in her fury at his voyeurism, she magically made his hounds rabid and they savaged their master. This version is presumably a Roman slant on the tale, given that Diana is the Roman name for the Goddess who, in Greek mythology, was called Artemis. Most Greek versions I have come across describe a similar chain of events: Acteon chances upon Artemis bathing naked in a woodland pond; but they generally agree that she turned him into a stag, where upon his hounds attacked.
Curiously, despite no specific mention of rabies in any of the stories, the fact that Aceton’s hounds apparently attacked their master with a ‘wolf’s frenzy’, suggests to some that they may have been rabid. Regardless, rabies was known in Aristotle’s time and around the 4th Century BCE he described how dogs suffered from a madness that caused them to become irritable. Crucially, Aristotle noted how “all animals they bite become diseased”, thereby recognising that the disease could be passed on in the bite of an infected animal. Fortunately, a great deal has changed since Aristotle’s time and not only do we now know what rabies is, we can modify its genetic code to produce an effective vaccine.
Knowing your enemy
Rabies is a highly infectious disease caused by a virus; specifically, a negative-strand RNA virus. There 12 recognised species of the rabies virus, and they’re all members of the same Lyssavirus genus, from the Greek lyssa, meaning ‘frenzy’ or ‘madness’ and the Latin virus, meaning ‘poison’. All mammals are susceptible to one species or another. The virus has a characteristic bullet-shaped appearance and is surrounded by a membrane envelope with surface projections. The virus particles (called virions) are tiny, only 180 nanometres long by 75 nanometres wide – to put it another way, if you laid them end-to-end, there would be just over 55,500 in one centimetre or 141,000 in an inch. Rabies, like all viruses, requires a live host cell in which to replicate. The virus is very resistant to low and freezing temperatures, but susceptible to heat, ultraviolet radiation and common detergents; it survives only a few hours at room temperature.

Rabies is generally divided into two broad ‘groups’ based on their primary hosts. Epidemics that sweep through humans and domestic animals are referred to as classical rabies, while those that occur in wild animals are called sylvatic rabies. It should be noted that these categories are based only upon the main reservoir for the virus; the virus causing the infection is the same. In a 1995 paper to Seminars in Virology, a team at the Center for Disease Control in Atlanta described how virus variants tend to circulate within specific species groups. In other words, we could identify ‘fox rabies’, ‘coyote rabies’, ‘raccoon rabies’ and so forth. This has been interpreted by some as meaning that rabies viruses are species-specific (i.e. fox rabies isn’t communicable to, for example, dogs), but this is not technically the case. As Nardus Mollentze and his colleagues point out in their 2014 paper to Current Opinion in Virology, what actually happens is that the rabies virus variants typically do not jump from one maintenance host to another. A maintenance host is an individual that can shed virions, thereby passing the virus to someone else.
Spill-over, where one infected species transmits the virus to a different species, is well-known but the virus generally isn’t sustained in the second species. When dogs, for example, become infected with the virus they survive in an infectious state during which they shed rabies virions in their saliva. During this time, they’re aggressive and can pass the virus to other dogs during fights; these other dogs will subsequently pass the virus to more dogs and hence the virus propagates. When a rabid dog bites, say, a human, the virus doesn’t behave in the same way and the human doesn’t shed virions, so the virus doesn’t naturally pass human to human. The virus may be more generalist, however, and may circulate within a canid pool, allowing it to pass between the local dog and fox populations. Interestingly, bat rabies is often considered distinct from sylvatic rabies because it does not appear to readily infect other terrestrial mammals, although it can kill humans.
Overall, the picture is complicated and there are still aspects of the virus we do not fully understand. There are studies suggesting, for example, that ‘fox rabies’ doesn’t kill striped skunks (Mephitis mephitis), while ‘skunk rabies’ can kill foxes, but not raccoons (Procyon lotor). Such variations appear to be a reflection of which parts of the brain the virus variants are able to infect, but more data are needed.
Method of infection
The most common method of infection is the virus entering the body through a bite from an infected animal. The virus cannot be transmitted through intact skin but it can enter the body across mucous membranes such as the nose, mouth and eyes; sufficiently prolonged contact must be made, however, making it an unlikely source of infection for most species. Once in the body, the virus incubates for between two and eight weeks on average, although up to four months has been reported. How long symptoms take to develop depends on where the victim was bitten and thus how long it takes for the virus to move into the central nervous system (CNS). In order to enter the CNS it must first build up sufficient numbers (concentration), which it does by replicating in striated muscle at or near the bite site.
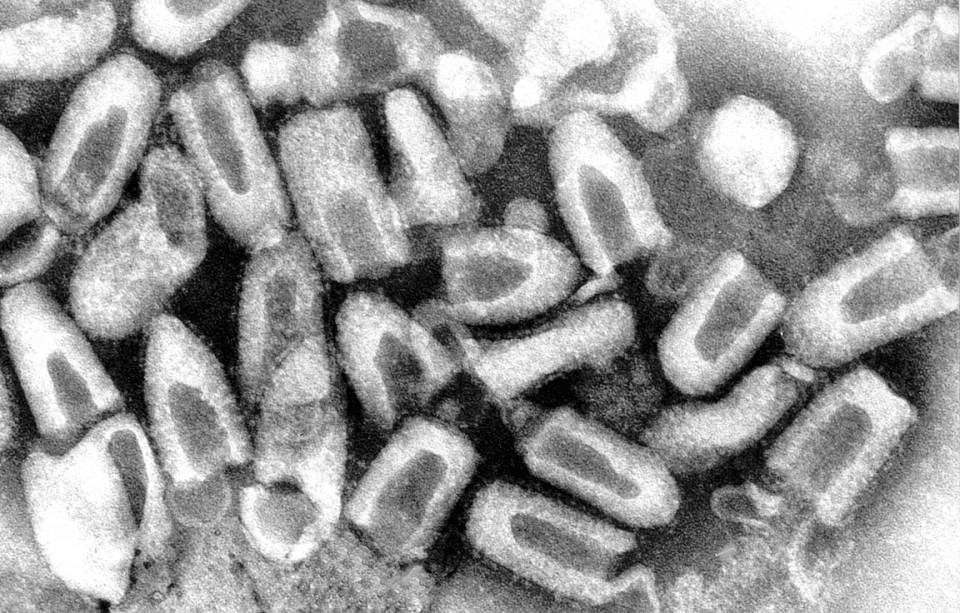
The virus’s main target is the brain, which is accessed via the CNS and the virus moves through the muscle fibres trying to avoid detection by the host’s immune system. The virus binds to receptors entering nerve cells and is delivered from the peripheral nervous system into the CNS in the axoplasm (nerve fluid). Once in the CNS it moves along nerves to a suitable spot within the brain where it can reproduce. The virus replicates in the brain, producing infectious virus particles (virions) that pass back down nerves to organs, particularly the salivary glands which allow its transmission through a bite. For an excellent, in-depth discussion of the virus and its behaviour inside the body, the reader is directed to Alex Wandeler’s 1980 review in Biogeographica.
Symptoms of the disease
The site of virus replication within the brain is important because it determines the ‘form’ of rabies that an animal contracts. If the virus establishes itself in the limbic system, the inner part of the brain, the victim is subject to agonising muscle spasms that prevent them from drinking; any attempt at swallowing water, even saliva, can trigger muscle spasms and the victim may come to fear water, a condition known as hydrophobia. The victim becomes highly aggressive, biting without provocation, and hence this is often referred to as the furious condition. If the virus replicates in the neocortex, the brain’s outer layer, the animal becomes progressively paralysed, often to the point where it cannot bite, and this is known as the dumb condition. It is important to note that both classical and sylvatic rabies can be either furious or dumb in nature.
Whether or not a rabid animal is likely to bite seems also to depend on the species. Anecdotal evidence has long suggested that rabid foxes are more likely to bite humans than other rabid animals and, in a paper to the Journal of Wildlife Diseases in 2011, a team of American biologists suggest there is some foundation to this rumour. The team, headed by Kimberly Yousey-Hindes at the New York State Health Department, studied Human Rabies PEP surveillance data collected between 1997 and 2007; they found that Red foxes infected with raccoon rabies were more likely to bite people than infected raccoons were. Despite furious rabies being the more stereotypical form of the disease, infected animals travelling widely and attacking without provocation, the dumb condition is actually more common. Regardless of the condition, once the animal is symptomatic, death typically follows within a few days. If the wound is flushed and a rabies immune globulin vaccine administered within a day of infection, however, the patient has a high chance of recovery.

Other symptoms of the disease include seizures, “losing fear of humans”, disorientation and walking around in what the media often describe as a “zombie-like state”. It is important to note, however, that these are non-specific symptoms and exhibited individually have several potential causes other than rabies.
Rabies in Britain and Europe
Thankfully, Britain is currently free of rabies; but this has not always been the case. There are mentions of the virus in the UK back as far as 1026, although it wasn’t until much later, in 1793, that a plan for eradicating the disease (in the form of a total ban on importing dogs) was implemented. Less than 30 years later, a high-profile loss to the virus came when Charles Lennox, the fourth Duke of Richmond, died from rabies while on a tour of Canada. The circumstances around the Duke’s contraction of the virus are unclear. Some sources suggest the duke was bitten by a dog, while other, arguably more reliable writers, state it was from a bite he received to his hand from a tame fox. There is even one account suggesting he contracted the virus through a shaving cut when he was licked on his face by a tame fox. Either way, the unfortunate Duke developed hydrophobia and died in Quebec in 1819.
In 1897 the Victorians passed the General Rabies Order, which required dogs taken out in public to be muzzled and authorised the shooting of any un-muzzled or stray animals in certain districts. Despite classical rabies having been a historic problem in Britain, sylvatic rabies never became established. There were, however, two cases of rabies in park deer during the Victorian era: one in Barnsley during 1856; and a second in Richmond Park during 1886/87, when 257 Fallow deer (Dama dama) were infected and observed biting each other.
In 1886, rabies became a notifiable disease under the Contagious Diseases (Animals) Act, meaning that it was an offence not to report suspected cases to the authorities. Following years of strict stray dog control, the UK and Ireland were declared rabies free in 1902 and 1903, respectively, and there were no further outbreaks until 1918, when First World War servicemen smuggled rabid dogs back from the European war zone. The first outbreak was in Plymouth, but the disease quickly spread and eventually most of the south of England was affected. The disease was subsequently eradicated and Britain was once again rabies free by 1922, when compulsory six-month quarantine was introduced for dogs coming into the UK from abroad.

Between 1922 and 1969 there were 27 cases of rabies in the UK; most were in dogs and all were contained within quarantine. In 1969 and 1970, however, the last documented cases of rabies occurred in two dogs that had completed their quarantine period and, as a direct result, a compulsory rabies vaccination scheme for pet dogs and cats imported to the UK was introduced in 1971.
In 2000, following recommendations published in the 1998 Kennedy Report, the quarantine laws were revised and the Pet Travel Scheme (‘PETS’) was introduced; this allowed animals entering the UK from member countries to bypass quarantine, provided the animal holds a valid ‘Pet Passport’. The passport assures various stringent criteria are met, including having been successfully vaccinated against rabies and microchipped. As of February 2017, pets can be imported from all EU countries and 16 other ‘listed’ countries, including Switzerland and Iceland, provided the animal has a valid passport.
The last case of indigenous rabies in Britain (excluding several tourists who arrived with the disease) was in November 2002, when a bat worker was admitted to Dundee Hospital in Scotland suffering from rabies contracted through a bat bite; this was the first death from wildlife rabies in Britain since 1902. Studies on the Daubenton’s bat (Myotis daubentonii) carcass by the Department of the Environment Food and Rural Affairs (DEFRA) found European Bat Lyssavirus II present in its saliva. Despite the tragic consequences, this was not considered an epidemiologically significant event, because it did not spread. Indeed, while fatal to other mammals, it seems only bats can transmit bat rabies.

Britain has been fortunate; much of Europe and North America have not fared so well with rabies. The current epizootic (wildlife epidemic) in Europe started just south of Gdansk in Poland during 1939 and spread rapidly westwards at a rate of up to 60km (40 mi.) per year, reaching Germany in 1950, then Austria in 1966. The first Slovenian outbreak occurred in 1973 and the virus rapidly spread south. In Europe as a whole, more than 90% of the infected animals were (indeed, still are) Red foxes and by the start of the 1990s the rabies epizootic was at its peak, with most countries reporting infections.
With construction on the Channel Tunnel well underway, fears were raised over the possibility of rabid foxes coming over from France; at the start of the 1990s, France had reported more than 900 cases of fox rabies. Fortunately, these fears were unfounded, not least because of the various defences in place either end of the tunnel. In 1995, a year after the tunnel opened, France had reduced the number of rabies cases to only 13 and the disease was eradicated in 2003. In March 2014, a French man died from rabies believed to have been contracted while in Africa and, in May 2015 a dog died from rabies after having been illegally imported from Algeria. In December 2015, French authorities confirmed the country was again rabies-free.
North America has seen a shift from most rabies cases being primarily in domestic animals (classical) prior to 1960, to the majority (93% in 2014) being sylvatic in wild carnivores and bats. Foxes are a problem rabies vector (carrier) in the United States, but significantly less so than raccoons or skunks. According to the Center for Disease Control (CDC) in Atlanta, 30% of sylvatic rabies cases reported during 2014 were in raccoons, while 5% were in foxes; bats and skunks were also significant vectors, accounting for 30% and 26% of cases, respectively. In a paper to the journal Molecular Ecology in 2016, Karsten Hueffer and colleagues presented their data on the distribution of rabies variants in Alaska, which closely matches the population structure of Arctic foxes (Vulpes lagopus) in the region. The biologists concluded that, while Red foxes invariably have a role to play in maintenance of the virus, rabies in Alaska appears to be driven primarily by the Arctic fox.
The role of Mr. Tod

The behaviour of the rabies virus has been well studied in the fox and in his 1987 book, Running with the Fox, Macdonald explained that foxes succumb at a dose one-ten-thousandth of that required to infect a human. Once infected, the virus incubates in the fox for around 20 days as it travels into the brain where it replicates before migrating to the salivary glands; here it reaches high concentrations, such that a single millilitre could, theoretically, infect 34 million other foxes. Professor Macdonald noted that once the virus appears in the saliva the animal is infectious for six days and dies about four days, sometimes only 24 hours, later. In a review on fox behavioural ecology published in the Journal of Veterinary Medicine during 2003, German biologist Ad Vos noted how a fox can be infectious before any symptoms are evident:
“… so an apparently healthy fox that behaves completely normal can already transmit the virus.”
Contrary to the image I suspect most people would hold of a rabid fox, Macdonald pointed out that only around 11% develop the furious strain of the virus; a stance echoed by Vos who, in his 2003 paper, noted that extremely aggressive behaviour is not very common in rabid foxes because the majority suffer the apathetic (dumb) form, and transmission probably involves contact between a rabid ‘passive’ fox and a healthy individual. We now know that rabies does not have to be transferred in a bite, although this is by far the most common route; it can be transmitted via contact with infected tissue and urine. Indeed, some authors have suggested that foxes can catch bat rabies from eating, or even sniffing at, infected carcasses. Canada-based virologist Alex Wandeler, however, found that foxes were 100,000-times more resistant to oral infection with rabies than they were to injection, suggesting the virus is susceptible to destruction by gastric acid.
We know very little about how foxes infected with rabies behave. In 1985, pathologists Marc Artois and Michel Aubert reported the results of their study radio-tracking three rabid foxes in an area of Lorraine, north-east France. They found all three animals remained within their territory during the incubation period, but during the infectious phase they appeared disorientated and strayed several hundred metres beyond their normal range. All three died at the border of their own territory, although some had fresh wounds, suggesting they had been fighting. The foxes’ overall activity increased largely as a result of “numerous aimless day-time movements”, and the animals spent more time resting at the periphery of their ranges. It seems probable that this sudden increased movement and time spent at territory boundaries would increase contact with neighbouring foxes – it would have been interesting, had the animals lived longer, to see whether they invaded neighbouring territories.
We also have some data on how healthy foxes behave towards rabid animals, albeit from captive individuals. In a 1975 paper, CDC epidemiologist William Winkler described how one of his subjects turned away from an individual with dumb rabies, while another sniffed it all over; when the fox suffered seizures the healthy animals clearly tried to avoid contact. It is unclear from these observations whether the healthy fox could tell the other animal was sick, or whether turning away was a reaction to unexpected and frightening behaviour. Observations elsewhere describe foxes bringing food to injured members of their social group, suggesting they are aware when another fox is ill.
The social system of the fox plays a crucial role in controlling the spread of a rabies epidemic, but is a subject that has received little attention until relatively recently. Indeed, prior to the studies of Huw Gwyn Lloyd (DEFRA), David Macdonald (Oxford University) and Stephen Harris (Bristol University), little was known about the social behaviour of Red foxes in Britain. Early theories had these animals as antisocial loners, but many hours of pain-staking research in less than glamorous conditions slowly started to show a different, more social side, to Reynard. It quickly became apparent that, when trying to get a handle on how a disease will spread through a population, you need to understand that population.

Fox sociality is covered in reasonable detail in the main fox article, so I will only summarize it here. Generally-speaking, there are three main social ‘systems’ employed by foxes: pairs (adult male and female) predominate in upland areas where territories are large owing to the distribution of food; family groups (adult male and female with, usually female, cubs from previous litters) are more common in lowland, especially urban, areas where territories are smaller because food and shelter is relatively more abundant; finally foxes may spend some, or all, of their lives moving around rather than establishing a territory, these are the so-called itinerants. The formation of pairs or groups is not only an effect of food availability, it is also associated with density – in richer habitats, territories can be smaller, food is sufficient to support group formation and fox density is higher (i.e. there are more foxes in the area), while the opposite is true in poorer habitats. The rabies virus can spread faster through high density populations than low density ones.
Mathematical models suggest that rabies requires a critical threshold density (CTD) to spread; below this density an epizootic cannot be sustained. Most models put the CTD at around one fox per two sq-kilometres, although some early estimates from Europe suggest a much lower density is necessary and there must be less than one fox per five sq-kilometres (one every two sq-miles) to stop rabies spreading. In Britain, densities range from that single fox per five sq-km to more than two animals per sq-km. Neighbouring territory holders rarely seem to fight, but trespassers may be violently expelled, and there is a greater potential for increased contact (and hence fights) between foxes in high density populations. Given that the most likely route of transmission is through a bite or other open wound, fighting with an interloping rabid animal would seem almost certain to lead to infection. It should be noted that it is the territory owner who usually initiates the attack, so even though most foxes suffer dumb rabies, this does not preclude them from aggressive encounters.
Waves, peaks and troughs of infection
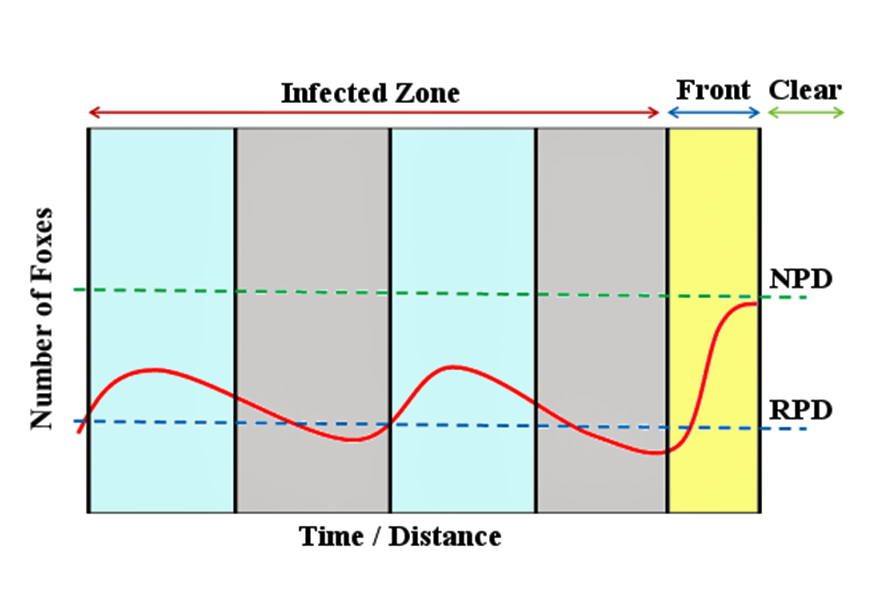
Rabies epizootics show distinctly seasonal and cyclical patterns in spread and intensity. In a 2006 review of wildlife rabies in Europe, Finish biologists Katja Holmala and Kaarina Kauhala described the spread of the virus within a population. When rabies enters a new area, there is a peak in reported cases among the main wildlife vector, although it has been estimated that fewer than 10% of cases are actually recorded. Thus, there is a ‘front’ of infection that spreads out into uninfected areas as a band, typically 40km to 100km (27–67 miles) wide.
As the front progresses the infected foxes in the zone behind it begin to succumb and it is estimated up to 80% of the resident foxes die, assuming around 20% avoid the infection. Such high mortality causes the fox population to crash below the CTD so the disease cannot spread – this leads to what is called a silent phase that typically lasts for two or three years. During the silent phase the incidence of the disease remains low, allowing the fox population to recover; as numbers increase they reach the CTD and the population becomes re-infected, probably from contact with rabid foxes moving out of nearby infected zones left as the ‘front’ advances. Thus, countries in the wake of the infectious zone tend to experience rabies epidemics that cycle between epizootic (infectious) and silent phases until the disease is eradicated.
We also see seasonal fluctuations in infection that are associated with fox biology and behaviour, with reported cases tending to show two peaks.
The first peak occurs in late winter and infection is predominantly among male foxes. Tracking studies have shown that this is when males trespass into neighbouring territories in search of vixens to mate with, bringing them into conflict with the territory owner and increasing the number of fights. Reports of rabies then settle down during the spring and early summer when pregnant females have given birth and adults confine most of their movements to their own range while raising the cubs.
The second peak is during the late summer and this time it’s the vixens that form the bulk of reported cases. In his fascinating 2003 review, Ad Vos pointed to research on reproductive stress in ungulates and suggested that this second peak may arise because the energetic costs of pregnancy and maternal care, coupled with the hormones released during birth and lactation that act to suppress the immune system, may make vixens more susceptible to infectious diseases.
In his review, Vos noted that cases of rabies in fox cubs are very rare and, where they do occur, are probably infections picked up from adult group members. Consequently, during the summer months, the juveniles aren’t a significant rabies risk because they remain within the core area of their parents’ range and don’t participate in territorial defence. Dispersal begins in August, peaking in October, and juveniles can then potentially spread the disease; they can certainly travel widely and this puts them into contact with other foxes.
Nonetheless, a radio-tracking study of dispersing and non-dispersing foxes in Bristol, by Tom Woollard and Stephen Harris, found no evidence that dispersing juveniles fought more, or were wounded more, than non-dispersers; an earlier study in Bristol did, however, find that dispersers had significantly lower life expectancy than non-dispersers. Overall, the role of dispersing foxes in the spread of rabies remains poorly understood and Vos concluded that it was ultimately territory holders that controlled the spread of the disease because they attacked intruders, regardless of whether the interloper was itself aggressive; this makes them extremely susceptible to becoming infected.
Kill or Cure?
Rabies is an old disease and an effective vaccine against it is a comparatively recent development. The traditional European view had always been, therefore, that rabies could be eradicated if sufficient foxes were killed. Indeed, in a brief article to the New Scientist during October 1990, Bristol University biologists Stephen Harris and Graham Smith discussed what would happen if sylvatic rabies ever came to Britain and how being ‘too sentimental’ could have disastrous consequences. The biologists presented a computer model showing how rabies might spread were it to arrive in Bristol under three scenarios: no control; vaccination; and culling.
No control
The model predicted that without any form of control, the disease would flare up quickly such that almost all the urban foxes in Bristol would be dead within three years and the virus would have reached the surrounding countryside. A vaccination campaign that reached (an optimistic) 70% of the fox population slowed the spread, but it still eventually breached the city and made it to the countryside. It transpires that only with an intensive cull (i.e. five culling operations at two-monthly intervals that killed 92% of the foxes) was it possible to eradicate the disease within one year. This approach has been widespread in both Europe and North America and when rabies hit Albert in 1952, it prompted a massive cull of wildlife with, among many other species, 105,000 coyotes and 50,000 foxes killed. So, is culling the best, indeed the only, option?

Culling foxes
The problem with widespread culling is that it is difficult to achieve the desired result. Decades of persecution through fur trapping, anti-rabies campaigns and more general campaigns to halt the spread of fox populations have generally had little success at reducing colonisation, decreasing numbers or wiping out rabies. Indeed, in some areas, rigorous culling has actually prolonged the epizootic when compared to similar areas in which there was little or no culling. There are, of course, some exceptions.
In Jutland, on the Danish peninsula, an intensive campaign of shooting and gassing foxes coupled with poisoning of feeding station baits with strychnine nitrate succeeded in eradicating rabies within two breeding seasons, on three separate occasions. Nonetheless, in Europe as a whole (and in most areas where fox control is practiced) culling has had little impact on numbers because the fox population is quick to respond with increased fecundity (see QA for more). Indeed, culling may cause more problems than it solves if territory holders are principally responsible for rabies transmission.
Culling sends the population into a state of flux; vacant territories arise as the owners die and these are rapidly taken over by foxes dispersing from elsewhere, which then potentially leads to more fighting as boundaries are re-established. If dispersers do turn out to play a role in rabies transmission, having a mobile population that is predominantly made up of juvenile dispersing foxes could make the situation worse still. Culling must also be long-term, keeping the population low for long enough for the disease to die out; this becomes less cost efficient as the number of rabies cases drops off.
Vaccinating foxes
If long-term culling is problematic, not to mention expensive and more difficult as fox numbers decline or animals become warier, is there an alternative? In recent years, vaccination appears to have succeeded where culling has failed. In a 1982 paper, David Macdonald and Philip Bacon presented their Merlewood fox rabies simulation model, which predicted that vaccination against rabies was a realistic alternative to culling provided sufficient bait-uptake could be achieved; they assumed 60% of the population took the baits. The basic idea of any vaccination campaign is to create a CTD using immunized animals. In other words, it essentially mimics heavy culling by ensuring so many foxes are immune to rabies that it cannot physically spread. To prevent re-infection, it is necessary to establish such ‘immune belts’ (i.e. areas where most, ideally all, foxes are inoculated) along borders.
A subsequent model by Oxford University mathematician James Murray and his colleagues, published in 1986, assessed the spread of rabies among foxes after an introduction near Southampton in southern England. The model predicted that the epizootic spreads out at a rate of up to 100 km (67.5 miles) per year, travelling fastest through central England where fox densities are highest. It covers the entire West Country in just over two years, Wales in just over three years and reaches Manchester in just under four years; this is followed by periodic ‘waves’ of the disease every six years. The mathematicians concluded from their model that, in order to halt the spread of the virus, it is necessary to establish “rabies breaks” (immune belts) 10-25km (7-17 miles) wide, in which at least 80% of the population were vaccinated, ahead of the advancing "front".
So, what is a vaccine? When a foreign body (a bacteria or virus, for example) enters our body, specialised cells in our blood called lymphocytes target it with the aim of destroying it. Mammals produce two types of lymphocytes: B-cells and T-cells. B-cells circulate in the blood looking for invaders (which we call antigens) and when they’ve been identified an antigen they produce proteins called antibodies that specifically bind to the invader. The T-cells are then attracted to the antibodies, attach to the pathogen and destroy it. More than this, when they bind with the antibody, the T-cells divide, creating more cells with the same antibody receptor; in this way, an "army" is built to ‘mop-up’ the invaders.
Now, as your body gets to the end of the fight and the infection is almost gone, far more of these antibodies and T-cells are being produced than are needed – this surplus remains in the blood. This means that if they see the invader again, they’re prepared and can target it straight away, hopefully before it gets out of hand. This process is called adaptive immunity and, although I have grossly oversimplified it, it’s important to understand the basics because vaccines take advantage of this system. Indeed, the goal of vaccination is to ‘train’ someone’s immune system to recognise a disease-causing agent, be it rabies or the ‘common cold’, by exposing it to a ‘harmless’ version. It’s a dry run that ends with the equivalent of wanted posters hanging all over town. This can be done in two main ways: you can inject a very low dose that produces only mild symptoms, but does no lasting harm; or you can introduce a version of the pathogen that has been disabled in some way, like you might wash or cook contaminated food before eating it.
Weakened viruses have long been used in vaccinations, and this is how French doctor Émile Roux developed and tested the first rabies vaccine with his colleague Louis Pasteur almost 150 years ago; he injected rabbits with rabies before killing them and extracting their spinal cords, which he ground down and injected into other rabbits. Many repeats of this process later, they had a weakened rabies virus that could be used as a vaccine. Pasteur began administering their vaccine in 1885 and, according to Alan Jackson’s chapter in the 2013 compendium Rabies: Scientific Basis of the Disease and its Management, in 1886 alone Pasteur vaccinated 350 people, only one of whom subsequently developed rabies.

Recent advances in biotechnology and genetic engineering meant that we could administer chemicals that essentially kill the virus, although this makes it less effective as a vaccine, or even disguise less dangerous viruses as rabies – a sheep in wolf’s clothing, if you like. We can even remove certain genes on the live virus to prevent it being infectious, akin to letting down the tyres on the getaway car. The late 1980s and early 90s saw the development of several vaccines against rabies and a flurry of laboratory studies assessing their effectiveness on captive foxes.
Most vaccines proved successful at providing immunity, although some trials with the Street Alabama Dufferin (SAD) ‘strain’ of the vaccine did appear to cause rabies in a few individuals as well as being fatal to several rodent species and skunks. In a 2009 paper, Thomas Müller and colleagues reported six cases of foxes from Germany and Austria that appeared to have contracted rabies from a SAD vaccine variant.
Subsequently, in a 2014 paper to the Journal of Wildlife Diseases, Peter Hostnik and his co-workers described a rabies infection in a young male fox, shot in the Slovenian village of Most na Soči during May 2012, that they identified as having been caused by consumption of a bait containing the SAD B19 vaccine strain. We don’t yet know whether foxes can pass this vaccine virus strain to healthy animals, although Hostnik and his team did find the virus in the saliva of the Slovenian fox. It must be noted, however, that cases of vaccine-induced rabies are exceptionally rare given the hundreds of thousands of foxes that have been vaccinated, and SAD is not the only vaccine used in baiting campaigns.
Interestingly, there have also been a couple of individuals that seroconverted (produced antibodies) of their own accord, thus recovering without being vaccinated. Quite how this natural immunity arose is unclear, but there is evidence that inoculated vixens may pass some of their anti-rabies antibodies to their cubs. Indeed, several studies have shown that rabies-neutralising antibodies are found in cubs born to orally-vaccinated vixens, although we’re still unsure how this happens. Writing in a 2003 paper to The Veterinary Record, Ad Vos and his colleagues failed to find any antibody transfer directly to the foetus (i.e. across the placenta) and suggest that the immunoglobulins may be delivered to the cubs in the colostrum.
Müller and his co-workers found that the antibody level in cubs was directly related to the amount in the mother, suggesting colostrum is a probable source. Whatever the method of transfer, it appears that these antibodies provide only short-term protection. Müller and his colleagues, in their 2002 paper to BMC Infectious Diseases, reported that the maternally-transferred rabies antibodies dropped to a level where they were no longer likely to offer any protection by about 23 days old.
In a 2003 paper to a Czech veterinary journal, Hostnik and colleagues at the University of Ljubljana in Slovenia found that vixens vaccinated with the Lysvulpen anti-rabies vaccine transferred antibodies to their cubs that persisted for about two months. Short-lived maternally-derived antibodies interfere with vaccines, so these findings have important consequences for vaccination programmes because they suggest any attempt to vaccinate cubs less than about eight weeks old will achieve little.
To catch (and vaccinate) a fox
As you might imagine, rounding up foxes and injecting them with a rabies vaccine is a rather impractical idea. Thus, an alternative strategy was needed; this came in the form of bait vaccines. The basic premise is to conceal the vaccine in something the fox is likely to eat and drop it where the fox is likely to find it. Unfortunately, the challenges of this approach are many-fold. The bait must be relatively cheap to manufacture, attractive to the fox (otherwise it won’t get eaten), must not react with the vaccine to deactivate it while also providing some protection from the local weather – some early vaccines rapidly deteriorated above 4C/39F). The vaccine capsule must be easily ruptured by the fox and yet not so fragile as to break and spill the vaccine when it hits the ground. Finally, the bait must require chewing; this ensures the vaccine comes in contact with the fox’s oropharyngeal mucosa (tonsils) and stimulates an immune response.

The first baiting trials were conducted in Switzerland during 1978, where rabies was tracked as it moved along the ‘stalks’ of Y-shaped valleys in the Alps. In his book, Running with the Fox, David Macdonald explained:
“At the entrance to one arm of each Y every effort was made to kill foxes, while at the other arm chicken heads loaded with oral rabies vaccine were scattered – the foxes ate the chicken heads and thereby inoculated themselves. In these trials rabies progressed up the valleys until the junction where it met the two types of ‘barrier’. There, the disease continued up the arm of the valley where foxes had been killed, but was stopped in its tracks by the barrier of healthy inoculated foxes.”
The success of the Swiss Alps trial spurred interest in a more widespread and coordinated vaccination campaign across Europe, but chicken heads were expensive so alternative bait was required. Trials on captive foxes led to the development of a bait that is widely-used today – it is a square or rectangular fishmeal polymer bait into which a polyethene sachet (similar to the condiment packets you get in restaurants) is placed, sometimes encased in an additional wax casing that prevents the sachet falling out when it’s air-dropped. The sachet contains about 1.5 ml of vaccine and tetracycline. Tetracycline is deposited in bony tissues, showing up as a fluorescent ring under UV light; researchers can take tooth sections from foxes submitted for rabies analysis and tell by the presence of the tetracycline ring that the animal had eaten the vaccine bait. In other words, tetracycline is used as a bait marker.
The sachet is ruptured when the fox bites it, allowing the vaccine to make contact with the lymphatic tissue in the throat. It is important that it makes contact in the mouth; if the fox swallows the bait whole it is useless, because the vaccine is deactivated by the stomach acid.
Captive studies by biologists in Germany during the early 1980s found that the first antibodies were present in the fox’s blood about two weeks after bait consumption and maximum protection was recorded a further two weeks later (i.e. one month after the bait was eaten). It appears that this protection is generally fairly long-lasting and, in a 1997 study, Marc Artois and colleagues found that immunized foxes failed to succumb to challenge (i.e. were still immune to the virus) 18 months later. It is currently unknown precisely how long the antibodies remain, although if we consider that few urban foxes appear to exceed two years old, it may potentially provide life-long protection.
So, is vaccination a magic bullet for rabies eradication? The short answer is no. The success rate is heavily dependent upon a bait uptake success of 60% or greater; if too few foxes get the bait it leaves a sufficiently large pool of unprotected animals through which the virus can spread. This explains why baiting campaigns are more successful during the autumn and winter, when food is less abundant and bait is more likely to be taken. Bait drops in the spring or summer are likely to be ignored, cached or brought back to the cubs who are either too small to eat it, or too young to benefit from the vaccine.

Low bait uptake is also the main reason why vaccination could be problematic in urban environments; trials at the University of Bristol suggest that bait uptake by their foxes was only around 40%, because there was so much other food around. It is conceivable that householders might be persuaded to swap the food they deliberately leave out for vaccine baits, but nobody knows how well this would work. Hence, in areas where bait is unlikely to be taken and manually vaccinating the population (i.e. trapping and injecting them with vaccine) is impracticable, multiple waves of heavy culling may be the only realistic solution, as distasteful as it may appear at first glance.
Modern vaccines, while highly efficient, are also expensive, which makes it difficult both for developing countries to afford them and for more well-off countries to justify protracted baiting campaigns. At some point, as the number of rabid foxes declines, rabies becomes an increasingly minor problem and the financial costs of continued bait drops become more difficult to justify. What effect this will end up having remains to be seen, but all the time neighbouring countries have epizootics there is a considerable threat of resurgence. Indeed, Greece, for example, was declared rabies-free in 1987, following an intensive campaign of dog vaccination. However, Greece shares land borders with Turkey, Former Yugoslav Republic of Macedonia, and Bulgaria, all of which have rabies in both wild and domestic animals.
Animals generally don’t respect human boundaries and in October 2012 a rabid fox was found wandering a village in Palaiokastro, 60 km (37 miles) from the Albanian border, showing signs of the virus. Resources were mobilised and a vaccination campaign began in October of the following year. Discussing the results of the vaccination drive in a 2016 paper to the journal Vaccine, Laskarina-Maria Korou and her colleagues note:
“It could be assumed that the significant reduction in the number of animal rabies cases following the first [oral vaccination effort] and the non detection of rabies cases in Greece, since May 2014, may be related at least in part, to the protective immunity developed in wildlife. The second vaccination campaign that too place in Greece during the autumn 2014 may also have contributed to this. However, according to the experience gained by other countries, rabies elimination requires the implementation of [oral vaccination] programs for many consecutive years.”